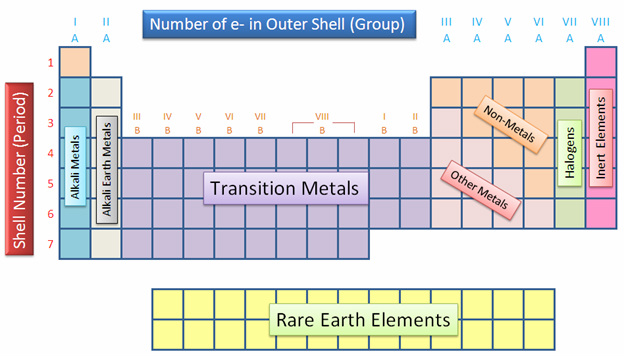
The Modern Periodic Table
The elements in the periodic table are arranged by group, period, valence electron, and orbitals.
Group:
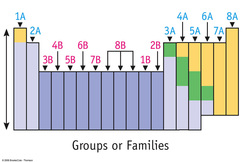
Groups, or family, are the vertical columns of the periodic table. There are 18 groups in the periodic table. Elements within a group have similar chemical and physical properties.
Period
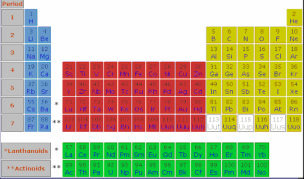
Periods are the horizontal rows of the periodic table. There
are 7 periods in the periodic table. The properties of the
elements vary as you move across the periodic table from
element to element.
are 7 periods in the periodic table. The properties of the
elements vary as you move across the periodic table from
element to element.
Valence Electrons:

Valence electrons are the electrons in the highest occupied
energy level of an element’s atoms. The elements in each
group in the periodic table behave similarly because they all
have the same number if valence electrons. The chemical
properties of an element are determined based on the number
of valence electrons. You can find the number of valence
electrons in an atom of the representative elements in the
periodic table just by looking at the group number it is in.
energy level of an element’s atoms. The elements in each
group in the periodic table behave similarly because they all
have the same number if valence electrons. The chemical
properties of an element are determined based on the number
of valence electrons. You can find the number of valence
electrons in an atom of the representative elements in the
periodic table just by looking at the group number it is in.
Orbitals:

The periodic table is divided into four orbitals: s, d, p, and f.
The s orbital consists of the 1st and 2nd groups of the
periodic table, also known as the alkali metals and alkali earth metals. The d orbital consists of the 3rd to 12th groups of the periodic table, also known as the transition metals. The p orbital consists of the 13th to 18th groups of the periodic table, also known as the other metals, metalloids, non-metals, halogens, and noble gases. The f orbital is the rare earth elements. These orbitals are used to figure out elements electron and noble gas configurations.
The s orbital consists of the 1st and 2nd groups of the
periodic table, also known as the alkali metals and alkali earth metals. The d orbital consists of the 3rd to 12th groups of the periodic table, also known as the transition metals. The p orbital consists of the 13th to 18th groups of the periodic table, also known as the other metals, metalloids, non-metals, halogens, and noble gases. The f orbital is the rare earth elements. These orbitals are used to figure out elements electron and noble gas configurations.